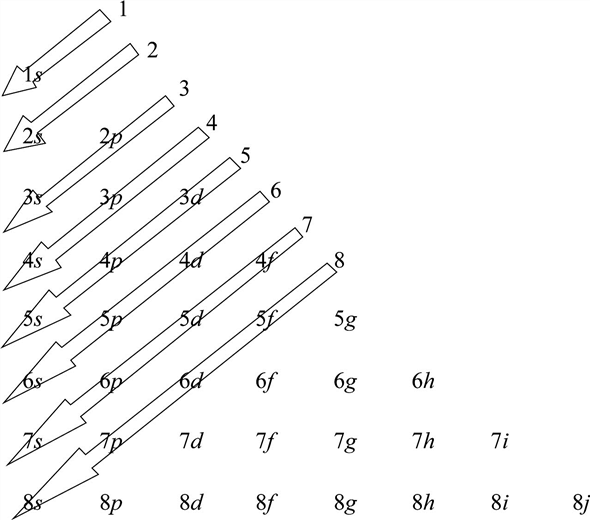
The electron will reside in the lower n subshell if two subshells have the same (n+l) value.For instance, the 3p subshell is referenced by n = 3 and l = 1. The values of n and l are discrete, such as n = 0, 1, 2,… and l = 0, 1, 2, and 3. According to the rising value of (n+l), the electrons fill up the subshell. The order of the subshells’ increasing energies is determined by the sum of n and l.They are the azimuthal quantum number l and the principal quantum number n. Four quantum numbers are present, two of which are related to the subshells. An electron’s state is indicated by a quantum number.Keep going in this manner through each subsequent energy level until all of the electrons have been distributed.Start by placing one electron in each of the three p orbitals in the second energy level (2p orbitals), and if there are still electrons available, go back and then add a second electron to complete the electron pairs.Insert the second two electrons into the s orbital of the second energy level (2s orbital).Add the first two electrons to the s orbital in the first energy level (1s orbital).Firstly, find out how many electrons are present in the atom.Therefore, the orbital penetration: s > p > d >f Steps for Aufbau Diagram Because the inner electrons shield the nuclear charge, orbitals fill in this manner. The ground state of the heaviest atom known, Z = 118, does not contain any of the orbitals listed in parentheses. (2) Energy rises with an increase in n for the same values of n and l.įilling orbitals in the following order yields the following results:ġs, 2s, 2p, 3s, 3d, 4p, 5s, 4f, 5d, 6p, 7s, 5f, 6d, and 7p (8s, 5g, 6f, 7d, 8p, and 9s) On Madelung’s rule, the modern Aufbau principle is hence based. Klechkowski described Madelung’s rule’s theoretical underpinnings. The filling of atomic orbitals and electron configuration are described by Madelung’s rule.Ĭharles Janet first described Madelung’s rule or Klechkowski rule in 1929, and Erwin Madelung rediscovered it in 1936. The orbitals are filled with electrons in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, and so on.The orbital with the lower n value is said to have lower energy associated with it if two orbitals have equal (n+l) values. Lower orbital energies are correlated with lower (n+l) values.The (n+l) rule, which states that the orbital’s energy level is determined by the sum of its principal and azimuthal quantum numbers, can be used to determine the order in which the energy of orbitals increases.This suggests that electrons only move into orbitals with higher energies once orbitals with lower energies are fully occupied. 
The Aufbau principle states that electrons first occupy orbitals with the lowest energies. 
The “circular orbits” outside the inner electrons have the highest angular momentum, but the s- and p-subshell orbits have high subshell eccentricity, which brings them closer to the nucleus and causes them to experience, on average, a less strongly screened nuclear charge. This was an early attempt to explain chemical properties in terms of physics by applying quantum mechanics to the characteristics of electrons.īefore quantum mechanics, electrons were thought to be in classical elliptical orbits according to the old quantum theory. In the early 1920s, Niels Bohr and Wolfgang Pauli developed the Aufbau principle, which is a key concept in the new quantum theory. The Aufbau principle states that atomic orbitals that are available and have the lowest energy levels are filled before those that have higher energy levels. Accordingly, electrons are inserted into atomic orbitals in ascending order of orbital energy. The Aufbau principle thus specifies how electrons are positioned in an atom’s ground state atomic orbitals. The Aufbau Principle contains historical predictions about the order in which they will fill. There are four different types of subshell shapes and seven different energy levels that electrons can occupy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
